VIDAS® D-DIMER EXCLUSION™ II
The power to exclude deep vein thrombosis and pulmonary embolism.
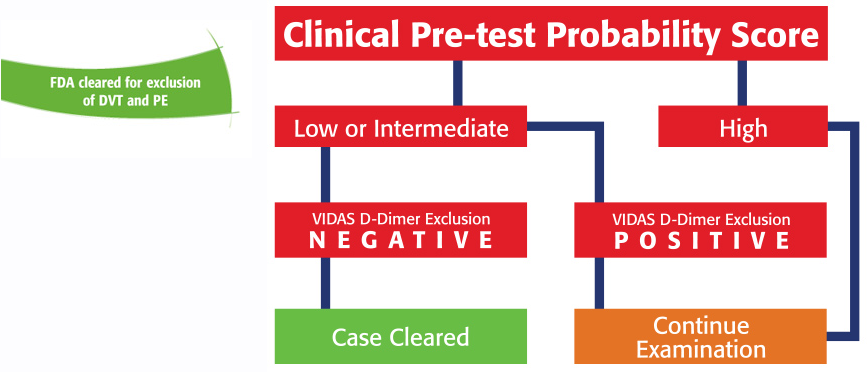
- FDA-cleared for exclusion of DVT and PE
- Rapid, automated ELISA
- >99% NPV in more than 50 peer-reviewed studies including >11,000 patients
VIDAS D-DIMER EXCLUSION II Overview
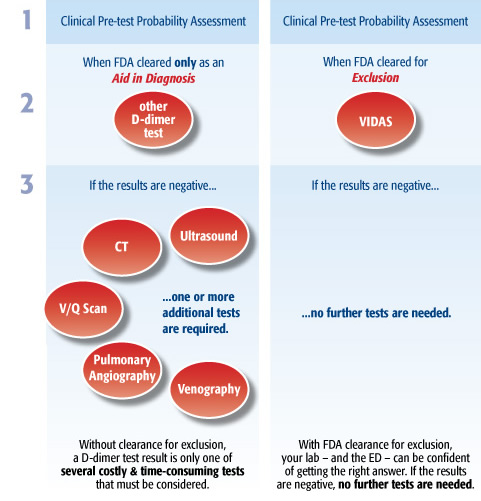
Your laboratory serves a crucial role by providing the ED with the information it needs to make accurate, fast, and safe diagnosis of patients with possible symptoms of PE or DVT. VIDAS D-DIMER EXCLUSION II provides confident exclusion of PE or DVT with a single test. With an exclusion strategy, ED physicians rely on D-dimer tests to rule out PE and DVT.
When used in conjunction with a clinical Pre-Test Probability model (PTP), VIDAS D-DIMER EXCLUSION II allows physicians to exclude deep vein thrombosis (DVT) and pulmonary embolism (PE) when a negative result is obtained. Because VIDAS D-DIMER EXCLUSION II is FDA-cleared for exclusion, no further tests are needed if the results are negative. VIDAS D-DIMER EXCLUSION II provides >99% NPV for all patients, even patients who have been determined to be at a medium risk for pulmonary embolism.
Just One Test. A Clear Answer. The Right Choice.
- The most referenced test for the exclusion of Venous Thromboembolism (VTE)
- A proven ELISA technique
- Expert validation in extensive prospective management clinical studies
- First FDA cleared assay for exclusion of Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) for outpatients with signs and symptoms of DVT and PE
- Rapid turn around time to support emergency medicine needs
- Educational Program Available on bioMérieux University: D-Dimer Testing: New CLSI Guidelines and Implications for Your Lab
About Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE)
Watch the video:
Disclaimer: All VIDAS instruments and assays are to be run by laboratory personnel in a laboratory certified under CLIA or by a CLIA approved State laboratory program. All personnel tasked with operating the VIDAS systems should be qualified and undergo a yearly competency assessment as defined in 42 CFR 493 subpart M “Personnel for Nonwaived Tests."
VIDAS D-DIMER EXCLUSION II Features
D-DIMER EXCLUSION in a Single Test
- First D-dimer test cleared by the FDA for exclusion of PE and DVT
- Offers >99% NPV for all patients-- even those presenting with medium risk for the most serious condition, Pulmonary Embolism
- Quickly and confidently rule out more than 1/3 of low to moderate risk outpatients who are presenting potential symptoms of pulmonary embolism or deep vein thrombosis
- Rapid, automated, enzyme-linked immunosorbent assay (ELISA)
- Results in less than 1 hour
- Ability to close patient cases and save resources when the test results are negative
- Help improve effiency in emergency departments
VIDAS D-DIMER EXCLUSION II Single Test Brochure
Disclaimer: All VIDAS instruments and assays are to be run by laboratory personnel in a laboratory certified under CLIA or by a CLIA approved State laboratory program. All personnel tasked with operating the VIDAS systems should be qualified and undergo a yearly competency assessment as defined in 42 CFR 493 subpart M “Personnel for Nonwaived Tests."
VIDAS D-DIMER EXCLUSION II Specs
| Code | DEXII |
| Reference | 30455-01 |
| Tests/Kit | 60 |
| Sample Type | Plasma (Citrate)/200µl |
| Sample Volume | 200ul |
| Calibrations | 1 level (included) every 28 days |
| Time to Result | 20 minutes |
| Results | Quantitative; Cut-off = 500ng/mL (FEU) |
The VIDAS D-DIMER EXCLUSION II assay is compatible with the following VIDAS instruments:
Disclaimer: All VIDAS instruments and assays are to be run by laboratory personnel in a laboratory certified under CLIA or by a CLIA approved State laboratory program. All personnel tasked with operating the VIDAS systems should be qualified and undergo a yearly competency assessment as defined in 42 CFR 493 subpart M “Personnel for Nonwaived Tests."
VIDAS D-DIMER EXCLUSION II Uses
Your lab can use the first FDA-cleared test to rule out a diagnosis of PE and DVT. VIDAS provides an exclusion strategy based on proven, validated results.1,2,3
VIDAS D-DIMER EXCLUSION II is the first test indicated for use to safely exclude a diagnosis of DVT and PE in outpatients with suspected VTE, in conjunction with a clinical pretest probability (PTP) assessment model.
- A safe and clinically validated exclusion diagnosis strategy
- Over 30% of outpatients with suspected VTE can be ruled out safely
- Reduced need for time-consuming and invasive examination
- Provides ELISA results in under 1 hour
1. VIDAS D-DIMER EXCLUSION II Package Insert
2. Perrier A, et al. Non-invasive diagnosis of venous thromboembolism in outpatients. Lancet.1999; 353: 190-195.
3. Perrier A, et al. Diagnosing Pulmonary Embolism in Outpatients with Clinical Assessment, D-Dimer Measurement, Venous Ultrasound, and Helical Computed Tomography: A multicenter Management Study, American Journal of Medicine. 2004; 116: 291-299.
Disclaimer: All VIDAS instruments and assays are to be run by laboratory personnel in a laboratory certified under CLIA or by a CLIA approved State laboratory program. All personnel tasked with operating the VIDAS systems should be qualified and undergo a yearly competency assessment as defined in 42 CFR 493 subpart M “Personnel for Nonwaived Tests."
VIDAS D-DIMER EXCLUSION II Webinars
VIDAS D-DIMER EXCLUSION II: Excluding a diagnosis of DVT and PE in outpatients with suspected VTE
This webinar focuses on the clinical use of the biomarker D-Dimer and as appropriate the assay VIDAS D-DIMER EXCLUSION II. The program includes clinical utility and an approach to exclude a diagnosis of DVT and PE in outpatients with suspected VTE.
Wednesday, June 8, 2016
Joseph Feldman, MD FACEP
Chairman, Emergency Services Emergency & Trauma Center
Hackensack University Medical Center
Meet the Speaker
Joseph Feldman, M.D, FACEPChairman, Emergency Services, Emergency & Trauma Center, Hackensack University Medical Center
Dr. Joseph Feldman completed his Emergency Medicine Residency at the The Brooklyn Hospital Center and is Board Certified in Emergency Medicine. He is an attending physician and Chairman of Emergency Medicine at HackensackUMC, where he has worked since 1998.
VIDAS D-DIMER EXCLUSION II Publications
Diagnosing Pulmonary Embolism in Outpatients with Clinical Assessment, D-Dimer Measurement, Venous Ultrasound, and Helical Computed Tomography: A Multicenter Management Study
Arnaud Perrier, MD, et. al.
The American Journal of Medicine, March 2004.
Diagnostic Pathways in Acute Pulmonary Embolism: Recommendations of the PIOPED II Investigators
Paul D. Stein, MD, et. al.
The American Journal of Medicine, December 2006.
Disclaimer: All VIDAS instruments and assays are to be run by laboratory personnel in a laboratory certified under CLIA or by a CLIA approved State laboratory program. All personnel tasked with operating the VIDAS systems should be qualified and undergo a yearly competency assessment as defined in 42 CFR 493 subpart M “Personnel for Nonwaived Tests."
The inclusion of a website link or resource material for download not original to bioMérieux Inc. is not intended as an endorsement of any product, service or advertisement and bioMérieux does not guarantee the accuracy of content. When users leave the bioMérieux website through those links, they are subject to the privacy and security policies of the owners/sponsors of the outside website. bioMérieux is not responsible for transmissions users receive from linked websites.