VIDAS® Lyme IgG II and VIDAS® Lyme IgM II
Is your laboratory prepared for Lyme Disease?
- Incorporates invariable region IR6 known as C6 peptide (VIDAS Lyme IgG II only)
- Reduction in total immunoblots for second tier testing
- Reduction in costs associated with immunoblots
- High sensitivity and specificity
- Rapid results in just 27 minutes
For More
Information
VIDAS Lyme Overview
The NEW VIDAS Lyme assays allow for independent measurements of IgG and IgM antibodies to Borrelia burgdorferi. When used together, these assays provide better correlation to disease stage and more specific information for the clinician for better patient management.1
Sensitivity and specificity of these assays is achieved with the addition of innovative recombinant chimeric proteins (VIsE, DbpA, OspC) allowing detection of all main pathogenic Borrelia strains (sensu stricto, afzelii, garinii). The design of the chimeric protein constructs includes C6 (the VIsE invariant region IR6), which allows the VIDAS Lyme IgG II assay to provide high sensitivity and specificity and limit cross reactivity. The VIDAS Lyme IgG II assay has a low level of cross-reactivity to syphilis and other infectious disease, and also reduces the number of unnecessary confirmatory tests needed, resulting in cost reduction.2
Testing with the automated VIDAS platforms can be done in batch or random access modes. Total time to results is 27 minutes facilitating quick diagnostic decisions.
VIDAS Lyme IgG II and VIDAS Lyme IgM II, are first tier assays for the presumptive detection of human antibodies to Borrelia burgdorferi. As such, positive and equivocal specimens should be further tested with a Western Blot assay. Notice that the VIDAS Lyme IgG II assay does not have an equivocal zone and only positive results must be confirmed.
References:
1. VIDAS Lyme IgG and VIDAS Lyme IgM package inserts available at www.mybiomerieux.com.
2. Wormser GP, Levin A, Soman S, Adenikinju O, Longo MV, Branda JA. Comparative cost-effectiveness of the two-tiered testing strategies for serodiagnostics of Lyme disease with non-cutaneous manifestations. J Clin Microbiol. 2013;51(12):4045-4049. doi:10.1128/JCM.01853-13
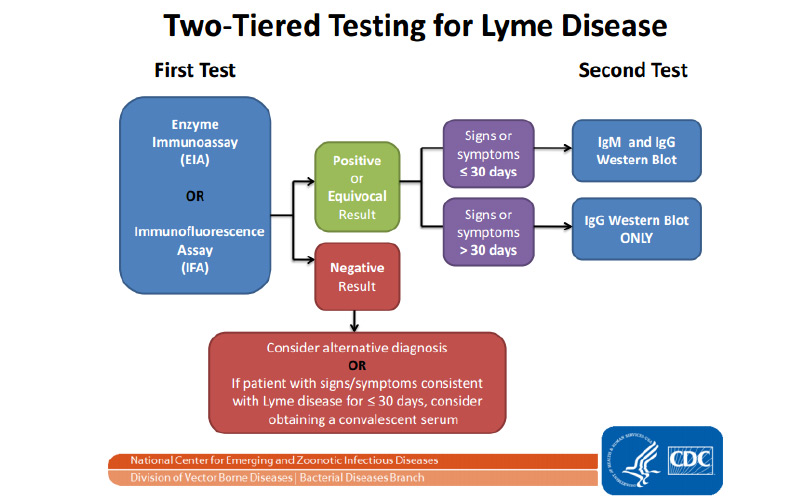
CDC's recommended Two-Tiered Testing for Lyme Disease
The CDC recommends a two-tier process for testing for Lyme disease. First, ELISA based assays are used mainly for screening followed by confirmation of positive or equivocal results by Western Blot (WB).
| IgM antibody | IgG antibody | Western Blot | Likely Interpretation |
| Positive | Positive | Positive | Likely Lyme Disease |
| Positive | Negative | Negative | Early infection or false positive |
| Negative | Positive | Positive | Late or previous infection |
| Negative | Negative | (usually not performed if IgM and IgG are negative) | No infection present, symptoms may be due to another cause or antibody levels too low to detect. |
VIDAS® Lyme Features
Differentiated serological profile
- Facilitates classification of Lyme borreliosis: early or late/chronic, active or immune
- Helps ensure patients receive rapid, optimized treatment depending on their stage of infection
- Potential to deduce information on disease progression (based on IgM and/or IgG positivity)1
Better patient management1 and only patients with Lyme borreliosis are treated
- Innovative recombinant chimeric proteins (VlsE, DbpA, OspC) allowing detection of all main pathogenic Borrelia strains (sensu stricto, afzelii, garinii). The design of the chimeric protein constructs includes C6 (the VIsE invariant region IR6), which allows the VIDAS Lyme IgG II assay to provide high sensitivity and specificity and limit cross reactivity
- Low level of cross-reaction (syphilis, other infectious diseases)
- “The LYT EIA had higher cross-reactivity when other diseases were tested (73% specificity) than the LYM/LYG EIAs (91% and 82% specificity when duration of illness was considered and not considered, respectively).”5
Easy result interpretation
- No equivocal zone for VIDAS Lyme IgG II results
- Avoid unnecessary patient anxiety and additional testing
Streamlined workflow, rapid results
- Results in 27 minutes
- Calibration only once every 28 days
- Ease-of-use
- Single-dose tests
Cost effective1,2
- Decreased number of total immunoblots that are needed for second-tier testing
- Reduction in costs associated with immunoblots
- Reduction in the level of specialized training that is required to properly perform an EIA as compared to immunoblotting
References:
1. Molins CR, Delorey MJ, Replogle A, Sexton C, Schriefer ME. Evaluation of bioMérieux's dissociated VIDAS Lyme IgM II™ (LYM) and IgG II™ (LYG) as a first-tier 6 diagnostic assay for Lyme disease. J Clin Microbiol. 2017;55(6):1698-1706. dio: 10.1128/JCM.02407-16
2. Wormser GP, Levin A, Soman S, Adenikinju O, Longo MV, Branda JA. Comparative Cost-Effectiveness of the Two-Tiered Testing Strategies for Serodiagnosis of Lyme Disease with Noncutaneous Manifestations. J Clin Microbiol. 2013;51(12):4045-4049. doi: 10.1128/JCM.01853-13
Technical Specifications
| VIDAS Lyme IgG II | VIDAS Lyme IgM II | |
| Code | LYG | LYM |
| Reference | 417401 | 416436 |
| Tests/kit | 60 tests | 60 tests |
| Sample type | Plasma, serum | Plasma, serum |
| Sample volume | 100 µl | 100 µl |
| Calibration | 1 level every 28 days | 1 level every 28 days |
| Time to result | 27 minutes | 27 minutes |
| Results | Qualitative; no equivocal range | Qualitative |
CDC Evaluation of a 2-Tier Lyme Disease EIA Testing Method and Potential Diagnostic Significance
This webinar, given by Dr. Ursula Lena Prisco, describes the potential diagnostic benefits of using the dissociated VIDAS Lyme IgM II and VIDAS Lyme IgG assays in a 2-tier EIA approach vs. the use of the standard EIA.western blot algorithm.