ETEST®
Improving Therapeutic Decisions
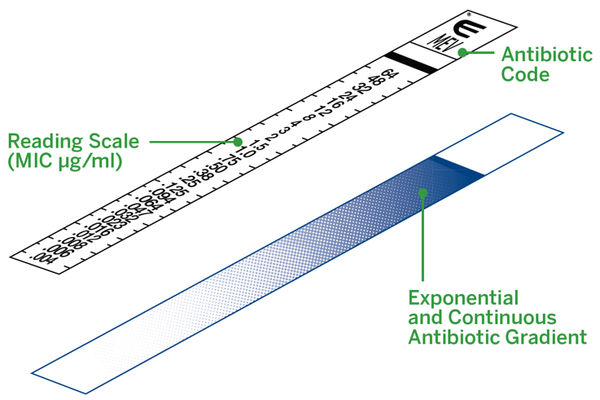
ETEST is a well-established method for Minimum Inhibitory Concentration (MIC) determinations in microbiology laboratories around the world. ETEST consists of a predefined gradient of antibiotic concentrations immobilized on a plastic strip and is used to determine the MIC of antibiotics and antifungal agents.
ETEST complements the VITEK® 2 automated system platform by offering a flexible and precise MIC solution to test additional antibiotics.
Overview
Quantitative MIC data is a prerequisite for the management of critical infections, including sepsis, especially among critical care patients. ETEST is a valuable diagnostic solution used to help guide and manage therapy when on-scale MICs are needed for treatment decisions. ETEST is part of an integrated ID/AST offer by bioMérieux to help complete your AST pathway and support your antimicrobial stewardship initiatives.
ETEST is also recognized as a flexible, cost-effective, and accurate tool used to detect or confirm emerging resistance and can provide faster availability of MICs for newer agents.
AST Powered By ETEST Gradient Technology
ETEST Complements the VITEK 2 System
ETEST complements the VITEK 2 system by offering a flexible and precise MIC solution to test non-fastidious organisms such as Enterobacterales, Pseudomonas, Staphylococcus, and Enterococcus species and fastidious organisms, such as anaerobes, N. gonorrhoeae, S. pneumoniae, Streptococcus, and Haemophilus species. The ETEST range offers more than 90 antimicrobial agents including resistance detection references.
ETEST Helps Fight Emerging Resistance
More than 2.8 million antibiotic-resistant infections occur in the U.S. each year.1 The emergence of resistant bacteria is a natural phenomenon that is exacerbated and accelerated by the inappropriate use of antibiotics.
ETEST provides a flexible diagnostic solution to test newer drugs and help fight emerging resistance. Ensuring patients get the right antimicrobial, the right dose, at the right time is the heart of antimicrobial stewardship
1. About Antibiotic Resistance. Centers for Disease Control and Prevention. https://www.cdc.gov/drugresistance/about.html. Published March 13, 2020. Accessed October 8, 2020.
Features & Specs
ETEST promotes the rational use of antibiotics by providing results to guide the therapy of individual patients and to validate empiric drug regimens. It is particularly helpful in determining choice and dosage of antimicrobials in patients with sterile site infections (i.e., endocarditis), severe nosocomial infections, chronic infections (i.e., cystic fibrosis) and immunosuppressed patients.
Four Simple Steps and Easy Interpretation*
 |
 |
 |
 |
||
*Refer to drug/bug specific package
Packaging Options
ETEST Strips are available to purchase in two different packaging options, allowing laboratories to order what fits their specific needs.
|
|
Single Pack Single packs contain one strip per package. The packaging is easy to open with one hand and a desiccant within each pack ensures stability. Available in 30 single packs.
|
|
Multi-Pack Strips are contained in cartridges which hold 10 strips. There are 10 cartridges per box for a total of 100 strips. There is an integrated desiccant in each cartridge and each box comes with a re-sealable lid for enhanced storage stability. This packaging is compatible with the SIMPLEX 76™ and the NEMA 88™. |
Support Instruments & Accessories
| RETRO C80™ | Mini Grip-It | NEMA C88™ | APPLICATOR SIMPLEX C76 |
 |
 |
 |
 |
| REF 559804 | REF 411200 | REF 559804 | REF 559802 |
| $1,225.00 | $36.55 | $1,202.00 | $7,037.65 |
|
ETEST Ceftazidime/Avibactam |
ETEST Ceftolozane/Tazobactam |
|
ETEST Delafloxacin |
ETEST Eravacycline |
|
ETEST Imipenem/Relebactam |
ETEST Meropenem/vaborbactam |
|
ETEST Piperacillin/Tazobactam |
ETEST Plazomicin |
|
ETEST Telavancin |
Downloads
ETEST Publications
Pseudomonas aeruginosa, and Acinetobacter baumannii:
Landman D, Bratu S, Kochar S, et al. Evolution of antimicrobial resistance among Pseudomonas aeruginosa, Acinetobacter baumannii and Klebsiella pneumoniae in Brooklyn, NY. J Antimicrob Chemother. 2007:60(1);78-82. doi: 10.1093/jac/dkm129
Pseudomonas aeruginosa and Enterobacteriaceae:
ASM Microbe 2017 - Performance of ceftolozane-tazobactam ETEST for antimicrobial susceptibility testing of Pseudomonas aeruginosa and Enterobacteriaceae. University of California, Los Angeles, CA. Poster #: Friday - 424.
Streptococci:
Rosser SJ, Alfa MJ, Hoban S, Kennedy J, Harding GK. ETEST versus agar dilution for antimicrobial susceptibility testing of Viridans group streptococci. J Clin Microbiol. 1999;37(1):26-30.
VRSA:
Tenover FC, Weigel LM, Appelbaum PC, et al. Vancomycin-resistant Staphylococcus aureus isolate from a patient in Pennsylvania. Antimicrob Agents Chemother. 2004;48(1):275-280. doi: 10.1128/AAC.48.1.275-280.2004
MIC for critical infections:
Potoski BA, Mangino JE, Goff DA. Clinical failures of linezolid and implications for the clinical microbiology laboratory. Emerg Infect Dis. 2002;8(12):1519-1520. doi: 10.3201/eid0812.020139
Cystic fibrosis:
Burns JL, Saiman L, Whittier S, et al. Comparison of agar diffusion methodologies for antimicrobial susceptibility testing of Pseudomonas aeruginosa isolates from cystic fibrosis patients. J Clin Microbiol. 2000;38(5):1818-1822.
Pneumococci:
Chaves F, Campelo C, Sanz F, Otero JR. Meningitis due to mixed infection with penicillin-resistant and penicillin-susceptible strains of Streptococcus pneumoniae. J Clin Microbiol. 2003;41(1);512-513. doi: 10.1128/JCM.41.1.512–513.2003
VRE:
Alam MR, Donabedian S, Brown W, et al. Heteroresistance to vancomycin in Enterococcus faecium. J Clin Microbiol. 2001;39(9):3379-3381. doi: 10.1128/JCM.39.9.3379-3381.2001
ETEST eLearning Opportunities
 Interactive ETEST Reading Quizzes Interactive ETEST Reading Quizzes |
Video: ETEST Multi-Pack Handling and Storage
PRN 059408 Rev 01.A