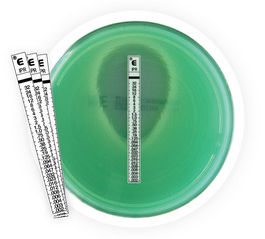
ETEST® IMIPENEM/RELEBACTAM
ETEST is a well-established method for Minimum Inhibitory Concentration (MIC) determinations in microbiology laboratories around the world. ETEST consists of a predefined gradient of antibiotic concentrations immobilized on a plastic strip and is used to determine the MIC of antibiotics and antifungal agents.
ETEST complements the VITEK® 2 automated system platform Learn more about ETEST.
Now FDA 510(k) Cleared!
ETEST IMIPENEM/RELEBACTAM
(IPR 0.002/4 - 32/4 µg/mL)
For More
Information
ETEST Imipenem/Relebactam is a quantitative technique for determining the susceptibility of Enterobacteriaceae. Imipenem/relebactam, brand name RECARBRIO™, is the combination of imipenem, a penem antibacterial and relebactam, a beta-lactamase inhibitor.1 ETEST Imipenem/Relebactam provides an additional diagnostic solution for multi-drug resistant Gram-negative organisms and helps clinicians guide and manage therapy.
Developed To Meet Emerging Resistance Challenges
ETEST Imipenem/Relebactam is used to assess susceptibility to imipenem/relebactam, an antimicrobial agent developed to treat urinary tract infections, including pyelonephritis (cUTI) and complicated intra-abdominal infections (cIAI).1
ETEST Imipenem/Relebactam is one of several ETEST strips developed as a part of an ongoing commitment to keep up-to-date with evolving microbiology needs. Other recent additions to the ETEST product line include ETEST Piperacillin/Tazobactam, ETEST Meropenem/Vaborbactam and ETEST Telavancin.
| Organism | Strains (N) |
% Essential Agreement (EA) |
% Category Agreement (CA) |
| Enterobacteriaceae | 447 | 95.8% | 98.1% |
Notes: EA = % of MIC values within ± 1 doubling dilution of the reference menthod.
Refer to Package Insert for additional performance data.
ETEST - MIC Testing
ETEST complements the VITEK 2 system by offering a flexible and precise MIC platform to test additional antibiotics. The ETEST range offers more than 90 antimicrobial agents including antifungals as well as antimicrobial resistant detection references.
ETEST Imipenem/Relebactam gradient strip is preformed and stable. It is a thin, inert, and non-porous plastic strip comprised of a predefined antibiotic gradient used to determine the MIC, in μg/mL, against microorganisms tested on agar media after overnight incubation. After 16-20 hours of incubation, you can determine MIC values for the organism. This strip supplements our ETEST product range.
Content of the Kit
Single Pack: 30 test strips
MIC range: 0.002/4 - 32/4 μg/mL
Ordering Contact Information
Customer Service: (800) 682-2666
To place and track orders, visit:
our eCommerce store
For Technical information, please visit: www.mybiomerieux.com.
References:
1. Recarbrio [package insert]. Whitehouse Station, NJ: Merck & Co, Inc.
RECARBRIO™ is a registered trademark of Merck & Co., Inc.